Classification of Medical Devices.
12/10/2021
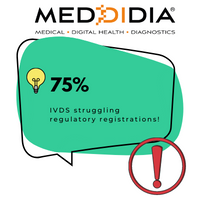
75% IVDs may Lack IVDR Certification!
21/9/2021
17/9/2021
8 Challenges Affecting Medical Devices
7/9/2021
Clinical Trials Information System (CTIS)
5/8/2021
Tech Supporting Medical Services!
3/8/2021
Med-Di-Dia's Presentation On Helix!
15/7/2021
Latest Guidance on UDI by USFDA
8/7/2021
Med-Di-Dia Takes Part in Innovation Workshop with ON Helix
5/7/2021
1/7/2021